This is due to the fact that with the increase in the size of ions, the distance between their nuclei increases. Why do lattice energy decreases down the group?Īs we move down the group, the lattice energy decreases. The atomic radius of atoms generally increases from top to bottom within a group.

The atomic radius of atoms generally decreases from left to right across a period. That means that the distance between the positive and negative ions is quite a lot less in MgO than in NaCl, and so the forces of attraction will be greater in MgO.Ītomic radius is determined as the distance between the nuclei of two identical atoms bonded together. Magnesium ions are smaller than sodium ions, and oxide ions are smaller than chloride ions. Why is the lattice energy of MgO higher than NaCl? These include By decreasing the atomic radius: So when we move across a period, the atomic radius of elements decrease wherein protons are pulling the electrons more and more. Generally, there are two main requirements for increasing lattice energy. Yes, the lattice energy increases as we move across a period. Does lattice energy increase across a period? What is born Haber cycle and its application?Ī Born–Haber cycle applies Hess’s law to calculate the lattice enthalpy by comparing the standard enthalpy change of formation of the ionic compound (from the elements) to the enthalpy required to make gaseous ions from the elements. Thus +2 or -2 ions will release more lattice energy than the +1 or -1 ions. Lattice energy increases with increase of charge on the ions because of their more attractive force between them. Why does lattice energy increase with charge? This definition causes the value for the lattice energy to always be positive, since this will always be an endothermic reaction. In one definition, the lattice energy is the energy required to break apart an ionic solid and convert its component atoms into gaseous ions. Bond making releases energy (exothermic step). Remember – bond breaking requires energy (endothermic step). There are no bonds to be broken, and so there is no endothermic step in hydration, only the release of energy when the bonds between the ions and the water are formed. Lattice energy and lattice enthalpy The formation of a crystal lattice is exothermic, i.e., the value of ΔHlattice is negative because it corresponds to the coalescing of infinitely separated gaseous ions in vacuum to form the ionic lattice. Is lattice formation exothermic or endothermic? +12891.5 kJ/mol What is the lattice energy of NaBr? Calculate the heat of formation of the Al +3 gasous ion. Given: Lattice energy =5376 kj/mol change in heat of formation = 326 kj/mol change of heat of formation = 122 kj/mol change of heat of formation = -233 kj/mol change in heat of formation = -704 kj/mol. How do you find the lattice energy of AlCl3? Question: Aluminum chloride, AlCl3, has a lattice energy of -5492 kJ/mol.

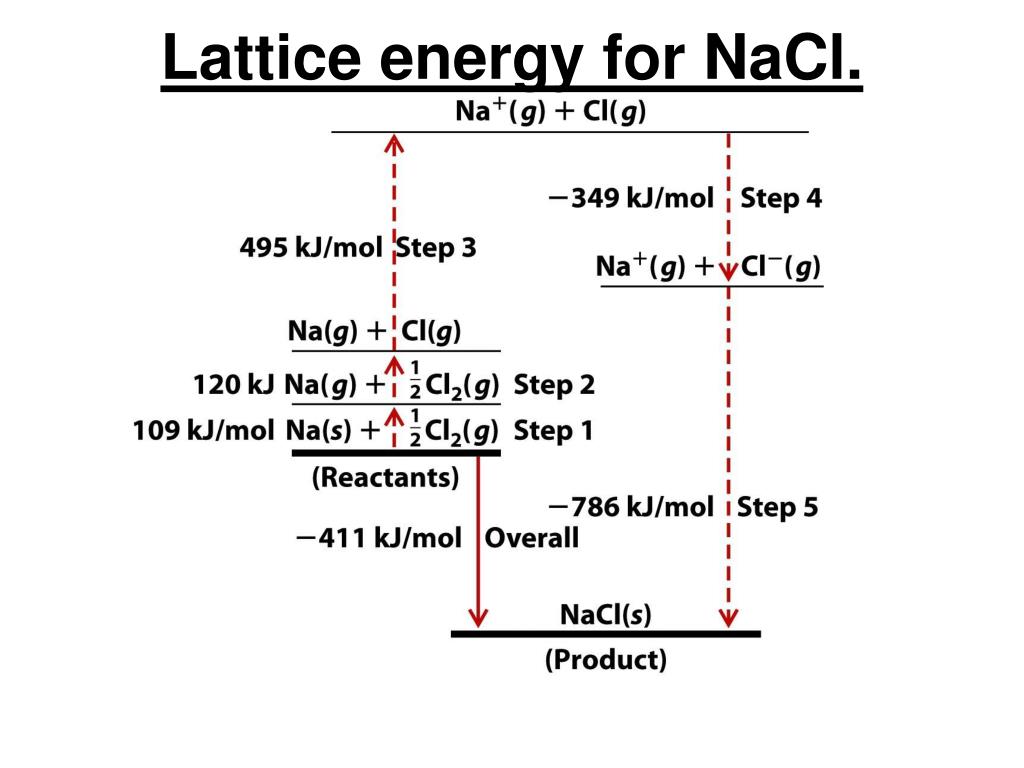
As the positive charge on Al is higher than Na, therefore the bond formed between Al-Cl would be stronger than Na-Cl, which is the main reason behind its higher lattice energy. Out of NaCl and AlCl3, the lattice energy of AlCl3 is higher than the lattice energy of NaCl. Which has more lattice energy NaCl or AlCl3? As a result the lattice enthalpy of MgO ishigher than NaCl. Because of this the ionic interactions are stronger in MgO than in NaCl.

➡️In MgO, the ionic bond is stronger than in NaCl because of more charge on the ionic species in the former (Mg+2 and O2- as compared to Na+ and Cl-). Which has higher lattice energy NaCl or MgO? Which one has the highest lattice energy? The lattice energy of CaO(s) is –3460 kJ/mol the lattice energy of K2O is –2240 kJ/mol. then, lattice energy of CaO =2×2=4=4U since, charge on each ion in CaO is of magnitude 2. Lattice energy of NaCl, U=1×1=1 since, charge on both ion is of magnitude 1. Lattice energy is directly proportional to the product of charge on ions in a crystal and is inversely proportional to the size of ions. Which has more lattice energy CaO or NaCl? The ionic bond in CaO (-2, +2 ions) is stronger than that in NaCl (-1, +1 ions).
